Scientist Interview
Introduction: My artwork represents the difference between a good ApoE3 and a bad ApoE4 protein. As you can see, the difference between the two is small, but the change of how they act can be tremendous. The background of the piece is black to represent how much is still unknown about these proteins. I choose to make the proteins look very realistic because I was using a picture that Dr. Mahley gave to me. You can see the picture that I used as a guide in one of the attachments.
Summary: Robert Mahley, PhD, MD, is known worldwide for his studies of the effects of a protein mutation. He studies the difference between the ApoE3 and the ApoE4 proteins and is trying to identify how exactly the proteins are mutated. Robert currently works with Gladstone Institutes, at UCSF, to learn more about the difference between ApoE3 and ApoE4. Robert has published studies about how a mutation in this protein can have direct connections to the development of Alzheimer's Disease and Cardiovascular problems. Robert found that the difference between the good ApoE3 and the bad ApoE4 are small, but because the ApoE4 is an incorrect shape, the cell recognizes it as an intruder, and attacks it. Robert has published many studies on how this mutation is the most direct genetic factor in the development of Alzheimer’s Disease and Cardiovascular problems that we know of today. He estimates that 65 - 80% of all Alzheimer’s patients have ApoE4. At Gladstone, he is working to come up with a drug that can actually use a ‘protein corrector’ to change an ApoE4 into an ApoE3.
When Robert is not directly involved in the lab work for his protein correctors, he spends most of his time trying to get funding to further his research. Robert tells me that he spends most of his day doing administrative tasks, but he has the most fun when he is spending time with his lab staff, going over experimental results. In recent years, Robert has transitioned his work from studying the ApoE3/ApoE4 proteins’ affect on the cardiovascular system to how those proteins affect the neurological system. Robert’s has huge goals for himself and his lab, he tells me “It is a never-ending story of discovery, with the goal of curing people with cardiovascular and neurological diseases.”
Ellie Holtaway: Your name, position, organization/laboratory/hospital.
Robert W. Mahley:
Name: Robert W. Mahley, M.D., Ph.D.
Position: Senior Investigator, Gladstone Institute of Cardiovascular Disease
Senior Investigator, Gladstone Institute of Neurological Disease
Organization: The J. David Gladstone Institutes
Position: Professor of Pathology and Medicine
Organization: University of California, San Francisco
My primary affiliation is with the Gladstone Institutes, which is an independent and nonprofit biomedical-research organization dedicated to accelerating the pace of scientific discovery and innovation to prevent, treat and cure cardiovascular, viral and neurological diseases. Gladstone is affiliated with the University of California, San Francisco.
Ellie: How did you come to research the structure and function of apolipoprotein (apo) E and it’s affects the cardiovascular system.?
Robert: I am attaching several documents that will give you background on what first sparked my interest in science (PNAS profile) and where my research has led me (from studying apoE in cardiovascular disease to new research on its role in Alzheimer's disease and other neurodegenerative diseases; also check out our website: http://gladstoneinstitutes.org/scientist/mahley and http://gladstoneinstitutes.org/node/11371)
Ellie: Can you tell me a little about your current work and its implications?
Robert:
Alzheimer’s Disease and Traumatic Brain Injury:
A Common Link and a Reason for Hope
FINDING THE CAUSE of Alzheimer’s disease is one of the greatest challenges facing modern medicine. Already, more than five million people in the United States are struggling with Alzheimer’s, incurring more than $200 billion in healthcare costs. And without a therapeutic breakthrough, the number of Americans with the disease is expected to more than the extensive damage to the brain is starkly evident in autopsy photos. The Alzheimer’s brain image on the right shows major parts of the brain that have deteriorated and died. Remarkably, the mechanism behind Alzheimer’s is the same as the one responsible for numerous other neuro-
Scientists now recognize apoE4 as the strongest genetic risk factor for Alzheimer’s disease.
The leading hypothesis about the cause of the disease has involved a plaque-like substance, called amyloid, that accumulates in and destroys brain cells. Unfortunately, a series of anti- amyloid drug tests have all failed to slow the progression of the disease—or alleviate its symptoms. The good news is that—thanks to four decades of research by Gladstone scientists into a protein called apoE—we now have a Plan B. After first establishing the link between apoE and heart disease, Gladstone scientists saw of apoE—known as apoE4—and the kinds of severe neurological damage seen in Alzheimer’s. As the pace of this research accelerated, the link between apoE4 and Alzheimer’s became unmistakable. Scientists now recognize apoE4 as the strongest genetic risk factor for Alzheimer’s.
That alone would be a dramatic scientific advancement. But remark- ably, apoE4 is not only implicated in Alzheimer’s. There is abundant evidence that it also plays a role in other neurodegenerative disorders. Traumatic brain injuries—the sort suffered by combat soldiers, pro- fessional athletes and car-accident victims—as well as multiple sclerosis, Parkinson’s disease and strokes are all linked to apoE4.
Not only have Gladstone scientists determined exactly how apoE4 wreaks havoc on brain cells, they have also identified damage from occurring in the first place. The science is rapidly falling into place. The hold-up? Funds to begin putting these research hypotheses to the test. With proper funding, human trials of these treatments could begin in as little as two years.
ApoE and Brain Damage Living cells make thousands of proteins, each with a specific role in keeping an organism alive. One instance, once apoE4 is produced, the cell detects its abnormal shape and considers it an intruder. In an attempt to defend itself, the cell then attacks apoE4 with a special molecule, called a protease, that cuts the protein into smaller—and toxic—fragments. Having nowhere to go, these hacked-off bits of apoE4 float protein, known as apoE, is usually made in brain cells, or neurons that are injured. Normally, apoE repairs neurons that have been injured— for example, by a concussion. ApoE does this by transporting first aid—in the form of special fat molecules such as cholesterol—to the cell’s exterior lining.
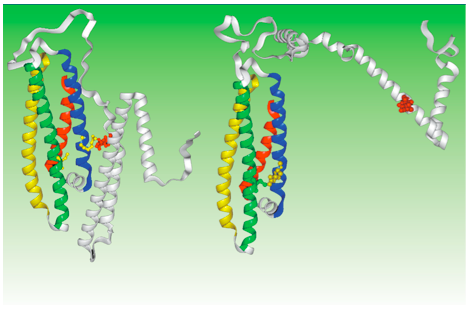
*See Image 1*
The difference between apoE3 and apoE4 is very small but that small difference has a huge effect on the structure and function of the two molecules.
ApoE4, because of its improper shape, gets stuck inside the cell. And that’s where the problems begin.
But there is a problem with apoE: It actually comes in several forms, including one that repairs cells (apoE3) and another that can destroy them (apoE4). And this form is not rare—as many as one in four people are predisposed to make apoE4. There is only one chemical difference between the “good” and “bad” forms of apoE. But the end result is two proteins with dramatically different shapes that behave in dramatically different ways. ApoE3 is transported to the outside of the neuron, where it does its repair work. But apoE4, because of its improper shape, gets stuck inside the cell. And that’s where the problems begin. A cell is an elaborately tuned machine that senses when something is not right. For aimlessly around in the cell. They end up damaging many of the cell structures they slam into, harming the cell in several ways. The recipients of some of the worst damage are the mitochondria, tiny “power plants” that create the energy the cell needs to live. The injuries caused by apoE4 slowly poison the mitochondria, eventually shutting down the power plant and killing the cell.
A similarly harmful injury occurs to the cell’s “cytoskeletal” system, which is an intricate transportation network. Imagine the tracks of a busy railroad station being yanked apart and tangled up with each other; apoE4 creates the same sort of chaos inside a cell, creating what are known as fibrillary tangles. The result is the death of the brain cell—a process multiplied many millions of times and affecting entire regions of the brain. Because these parts of the brain are associated with some of our most important human functions, including memory and social interactions, the result is the dementia and isolation we see in Alzheimer’s patients.
Mounting Evidence that ApoE4 Is the Culprit
Gladstone scientists have been studying apoE for nearly 40 years, making it one of the best-studied proteins in the human body. All the tools of 21st century life sciences have been brought to bear on this effort, from super-high-resolution microscopes to advanced 3D computer graphics. We now know the exact genetic code responsible for making apoE and how the different forms of apoE arise. We can also track, step-by- step, the destruction apoE4 causes inside previously healthy neurons. And considerable evidence suggests that apoE4 is causing conditions such as Alzheimer’s, as opposed to being just correlated with them. For example, an estimated 65–80% of all Alzheimer’s. Considerable evidence suggests that apoE4 is causing conditions such as Alzheimer’s, as opposed to being just correlated with them. Traumatic brain injuries reveal that those with apoE4 have consistently poorer clinical outcomes than those without it. That means they are significantly more likely to develop neurological diseases, including Alzheimer’s and Parkinson’s. There is even more compelling evidence about apoE4 from laboratory animal models. Mice bred with apoE4 perform significantly worse on standard lab tests that measure learning and memory, compared to mice with apoE3. And postmortem brain examinations of mice with apoE4 show the same regions of dead, shriveled neurons that we see in Alzheimer’s patients.
Racing to a Cure
The goal of everything we do at Gladstone is to quickly transform laboratory insights into actual treatments for patients. This approach guides our work with apoE4. The mechanisms described here suggest specific therapies not only for Alzheimer’s, but also for a range of other neurological diseases. Currently, we are developing three potential approaches to therapies to prevent the damage caused by apoE4:
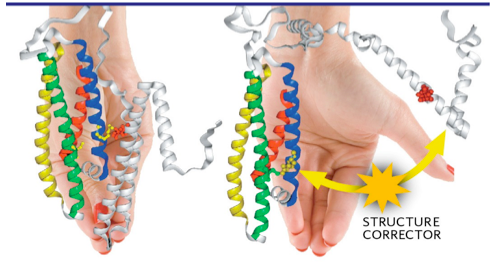
*See Image 2*
• A structure corrector. The difference between good and bad apoE involves a minute change in the structure, which has a profound effect on its function. We are actively exploring drugs that can repair this defect, patients have apoE4. In addition, studies of people who have suffered making the harmful apoE4 indistinguishable from the good apoE3. We believe this could prevent the cascade of cellular mayhem before it even begins.
At Gladstone, we’re working to develop drugs that can convert apoE4 to an apoE3-like structure.
• A protease inhibitor. An existing category of drugs, known as protease inhibitors, has played a key role in the fight against HIV/
WE ARE DEVELOPING CANDIDATE DRUGS AND NOW STAND AT A TIPPING POINT. AIDS. Now, new types of protease inhibitors may also be instrumental in inhibiting the detrimental effects of apoE4. We are working to develop a drug to prevent the in the disease process. And there is no reason to think we will not be able to include more than one approach in the same treatment regimen, creating an enhanced effect in which combined therapies are more effective than any single one.
We are developing candidate drugs for these three approaches and now stand at a tipping point. With proper funding, initial tests of an apoE4-related therapy could begin in as little as two years, suggesting that full-scale trials could be just three or four years away.
We expect the first human tests of our proposed therapies to focus on people who have suffered traumatic brain injuries, such as professional athletes or war veterans. If patients receiving therapy have better outcomes after six or eight months compared to those who didn’t receive the treatment, we could then expand the testing to other groups, including Alzheimer’s patients.
With this multipronged approach to stopping apoE4 from damaging the brain, we strongly believe we have a chance to prevent —and possibly reverse—the ravages of Alzheimer’s and other neurodegenerative conditions. We may not be able to change things for patients currently suffering with Alzheimer’s. But we hope and expect results that will help our children’s generation—and most certainly our children’s children.
TURNING GLADSTONE DISCOVERIES INTO TREATMENT
• Protective drugs. As a third line of treatment, we are exploring drugs that would shield both the mitochondria and the cytoskeletal
Ellie: We are interested in what your life is like as a researcher/clinician on a day to day basis. Is there something that you would like to share with the general public or community that cares about prevention, therapies and cures for the disease?
Robert: Science has enriched my life immensely. Even though I have to spend a considerable time doing administrative tasks, such as writing grants to support my research and writing manuscripts to inform other scientists about the work I do, the most fun times are meeting with my staff, going over the results of experiments, and planning new ones as one result leads to another and brings up new questions. It is a never-ending story of discovery, with the goal of curing people with cardiovascular and neurological diseases.
Ellie: As part of the 11th grade project, we are creating an art piece in studio art that I hope can serve to represent a piece of your work. Is there an image or idea that represents what you do that I can work towards to represent the important things that you do? If you don't have ideas now, we will be starting the artwork on Nov. 7th and would love to hear any ideas before then. Anything would help and would be amazing!
Robert: If you would like to use any of the images in the attached documents, please let me know, and I can send them to you. I hope you have a great time with your project. Feel free to contact me if you have additional questions.
Summary: Robert Mahley, PhD, MD, is known worldwide for his studies of the effects of a protein mutation. He studies the difference between the ApoE3 and the ApoE4 proteins and is trying to identify how exactly the proteins are mutated. Robert currently works with Gladstone Institutes, at UCSF, to learn more about the difference between ApoE3 and ApoE4. Robert has published studies about how a mutation in this protein can have direct connections to the development of Alzheimer's Disease and Cardiovascular problems. Robert found that the difference between the good ApoE3 and the bad ApoE4 are small, but because the ApoE4 is an incorrect shape, the cell recognizes it as an intruder, and attacks it. Robert has published many studies on how this mutation is the most direct genetic factor in the development of Alzheimer’s Disease and Cardiovascular problems that we know of today. He estimates that 65 - 80% of all Alzheimer’s patients have ApoE4. At Gladstone, he is working to come up with a drug that can actually use a ‘protein corrector’ to change an ApoE4 into an ApoE3.
When Robert is not directly involved in the lab work for his protein correctors, he spends most of his time trying to get funding to further his research. Robert tells me that he spends most of his day doing administrative tasks, but he has the most fun when he is spending time with his lab staff, going over experimental results. In recent years, Robert has transitioned his work from studying the ApoE3/ApoE4 proteins’ affect on the cardiovascular system to how those proteins affect the neurological system. Robert’s has huge goals for himself and his lab, he tells me “It is a never-ending story of discovery, with the goal of curing people with cardiovascular and neurological diseases.”
Ellie Holtaway: Your name, position, organization/laboratory/hospital.
Robert W. Mahley:
Name: Robert W. Mahley, M.D., Ph.D.
Position: Senior Investigator, Gladstone Institute of Cardiovascular Disease
Senior Investigator, Gladstone Institute of Neurological Disease
Organization: The J. David Gladstone Institutes
Position: Professor of Pathology and Medicine
Organization: University of California, San Francisco
My primary affiliation is with the Gladstone Institutes, which is an independent and nonprofit biomedical-research organization dedicated to accelerating the pace of scientific discovery and innovation to prevent, treat and cure cardiovascular, viral and neurological diseases. Gladstone is affiliated with the University of California, San Francisco.
Ellie: How did you come to research the structure and function of apolipoprotein (apo) E and it’s affects the cardiovascular system.?
Robert: I am attaching several documents that will give you background on what first sparked my interest in science (PNAS profile) and where my research has led me (from studying apoE in cardiovascular disease to new research on its role in Alzheimer's disease and other neurodegenerative diseases; also check out our website: http://gladstoneinstitutes.org/scientist/mahley and http://gladstoneinstitutes.org/node/11371)
Ellie: Can you tell me a little about your current work and its implications?
Robert:
Alzheimer’s Disease and Traumatic Brain Injury:
A Common Link and a Reason for Hope
FINDING THE CAUSE of Alzheimer’s disease is one of the greatest challenges facing modern medicine. Already, more than five million people in the United States are struggling with Alzheimer’s, incurring more than $200 billion in healthcare costs. And without a therapeutic breakthrough, the number of Americans with the disease is expected to more than the extensive damage to the brain is starkly evident in autopsy photos. The Alzheimer’s brain image on the right shows major parts of the brain that have deteriorated and died. Remarkably, the mechanism behind Alzheimer’s is the same as the one responsible for numerous other neuro-
Scientists now recognize apoE4 as the strongest genetic risk factor for Alzheimer’s disease.
The leading hypothesis about the cause of the disease has involved a plaque-like substance, called amyloid, that accumulates in and destroys brain cells. Unfortunately, a series of anti- amyloid drug tests have all failed to slow the progression of the disease—or alleviate its symptoms. The good news is that—thanks to four decades of research by Gladstone scientists into a protein called apoE—we now have a Plan B. After first establishing the link between apoE and heart disease, Gladstone scientists saw of apoE—known as apoE4—and the kinds of severe neurological damage seen in Alzheimer’s. As the pace of this research accelerated, the link between apoE4 and Alzheimer’s became unmistakable. Scientists now recognize apoE4 as the strongest genetic risk factor for Alzheimer’s.
That alone would be a dramatic scientific advancement. But remark- ably, apoE4 is not only implicated in Alzheimer’s. There is abundant evidence that it also plays a role in other neurodegenerative disorders. Traumatic brain injuries—the sort suffered by combat soldiers, pro- fessional athletes and car-accident victims—as well as multiple sclerosis, Parkinson’s disease and strokes are all linked to apoE4.
Not only have Gladstone scientists determined exactly how apoE4 wreaks havoc on brain cells, they have also identified damage from occurring in the first place. The science is rapidly falling into place. The hold-up? Funds to begin putting these research hypotheses to the test. With proper funding, human trials of these treatments could begin in as little as two years.
ApoE and Brain Damage Living cells make thousands of proteins, each with a specific role in keeping an organism alive. One instance, once apoE4 is produced, the cell detects its abnormal shape and considers it an intruder. In an attempt to defend itself, the cell then attacks apoE4 with a special molecule, called a protease, that cuts the protein into smaller—and toxic—fragments. Having nowhere to go, these hacked-off bits of apoE4 float protein, known as apoE, is usually made in brain cells, or neurons that are injured. Normally, apoE repairs neurons that have been injured— for example, by a concussion. ApoE does this by transporting first aid—in the form of special fat molecules such as cholesterol—to the cell’s exterior lining.
*See Image 1*
The difference between apoE3 and apoE4 is very small but that small difference has a huge effect on the structure and function of the two molecules.
ApoE4, because of its improper shape, gets stuck inside the cell. And that’s where the problems begin.
But there is a problem with apoE: It actually comes in several forms, including one that repairs cells (apoE3) and another that can destroy them (apoE4). And this form is not rare—as many as one in four people are predisposed to make apoE4. There is only one chemical difference between the “good” and “bad” forms of apoE. But the end result is two proteins with dramatically different shapes that behave in dramatically different ways. ApoE3 is transported to the outside of the neuron, where it does its repair work. But apoE4, because of its improper shape, gets stuck inside the cell. And that’s where the problems begin. A cell is an elaborately tuned machine that senses when something is not right. For aimlessly around in the cell. They end up damaging many of the cell structures they slam into, harming the cell in several ways. The recipients of some of the worst damage are the mitochondria, tiny “power plants” that create the energy the cell needs to live. The injuries caused by apoE4 slowly poison the mitochondria, eventually shutting down the power plant and killing the cell.
A similarly harmful injury occurs to the cell’s “cytoskeletal” system, which is an intricate transportation network. Imagine the tracks of a busy railroad station being yanked apart and tangled up with each other; apoE4 creates the same sort of chaos inside a cell, creating what are known as fibrillary tangles. The result is the death of the brain cell—a process multiplied many millions of times and affecting entire regions of the brain. Because these parts of the brain are associated with some of our most important human functions, including memory and social interactions, the result is the dementia and isolation we see in Alzheimer’s patients.
Mounting Evidence that ApoE4 Is the Culprit
Gladstone scientists have been studying apoE for nearly 40 years, making it one of the best-studied proteins in the human body. All the tools of 21st century life sciences have been brought to bear on this effort, from super-high-resolution microscopes to advanced 3D computer graphics. We now know the exact genetic code responsible for making apoE and how the different forms of apoE arise. We can also track, step-by- step, the destruction apoE4 causes inside previously healthy neurons. And considerable evidence suggests that apoE4 is causing conditions such as Alzheimer’s, as opposed to being just correlated with them. For example, an estimated 65–80% of all Alzheimer’s. Considerable evidence suggests that apoE4 is causing conditions such as Alzheimer’s, as opposed to being just correlated with them. Traumatic brain injuries reveal that those with apoE4 have consistently poorer clinical outcomes than those without it. That means they are significantly more likely to develop neurological diseases, including Alzheimer’s and Parkinson’s. There is even more compelling evidence about apoE4 from laboratory animal models. Mice bred with apoE4 perform significantly worse on standard lab tests that measure learning and memory, compared to mice with apoE3. And postmortem brain examinations of mice with apoE4 show the same regions of dead, shriveled neurons that we see in Alzheimer’s patients.
Racing to a Cure
The goal of everything we do at Gladstone is to quickly transform laboratory insights into actual treatments for patients. This approach guides our work with apoE4. The mechanisms described here suggest specific therapies not only for Alzheimer’s, but also for a range of other neurological diseases. Currently, we are developing three potential approaches to therapies to prevent the damage caused by apoE4:
*See Image 2*
• A structure corrector. The difference between good and bad apoE involves a minute change in the structure, which has a profound effect on its function. We are actively exploring drugs that can repair this defect, patients have apoE4. In addition, studies of people who have suffered making the harmful apoE4 indistinguishable from the good apoE3. We believe this could prevent the cascade of cellular mayhem before it even begins.
At Gladstone, we’re working to develop drugs that can convert apoE4 to an apoE3-like structure.
• A protease inhibitor. An existing category of drugs, known as protease inhibitors, has played a key role in the fight against HIV/
WE ARE DEVELOPING CANDIDATE DRUGS AND NOW STAND AT A TIPPING POINT. AIDS. Now, new types of protease inhibitors may also be instrumental in inhibiting the detrimental effects of apoE4. We are working to develop a drug to prevent the in the disease process. And there is no reason to think we will not be able to include more than one approach in the same treatment regimen, creating an enhanced effect in which combined therapies are more effective than any single one.
We are developing candidate drugs for these three approaches and now stand at a tipping point. With proper funding, initial tests of an apoE4-related therapy could begin in as little as two years, suggesting that full-scale trials could be just three or four years away.
We expect the first human tests of our proposed therapies to focus on people who have suffered traumatic brain injuries, such as professional athletes or war veterans. If patients receiving therapy have better outcomes after six or eight months compared to those who didn’t receive the treatment, we could then expand the testing to other groups, including Alzheimer’s patients.
With this multipronged approach to stopping apoE4 from damaging the brain, we strongly believe we have a chance to prevent —and possibly reverse—the ravages of Alzheimer’s and other neurodegenerative conditions. We may not be able to change things for patients currently suffering with Alzheimer’s. But we hope and expect results that will help our children’s generation—and most certainly our children’s children.
TURNING GLADSTONE DISCOVERIES INTO TREATMENT
• Protective drugs. As a third line of treatment, we are exploring drugs that would shield both the mitochondria and the cytoskeletal
Ellie: We are interested in what your life is like as a researcher/clinician on a day to day basis. Is there something that you would like to share with the general public or community that cares about prevention, therapies and cures for the disease?
Robert: Science has enriched my life immensely. Even though I have to spend a considerable time doing administrative tasks, such as writing grants to support my research and writing manuscripts to inform other scientists about the work I do, the most fun times are meeting with my staff, going over the results of experiments, and planning new ones as one result leads to another and brings up new questions. It is a never-ending story of discovery, with the goal of curing people with cardiovascular and neurological diseases.
Ellie: As part of the 11th grade project, we are creating an art piece in studio art that I hope can serve to represent a piece of your work. Is there an image or idea that represents what you do that I can work towards to represent the important things that you do? If you don't have ideas now, we will be starting the artwork on Nov. 7th and would love to hear any ideas before then. Anything would help and would be amazing!
Robert: If you would like to use any of the images in the attached documents, please let me know, and I can send them to you. I hope you have a great time with your project. Feel free to contact me if you have additional questions.